|
Lattice Energy = 147kJ/mol + 2188. Lattice Energy = Δ H o sub + I.E + Dissociating energy + E.A - ΔH f o( MgCl 2 ) ΔH f o (Mg Cl 2 )= Δ H o sub + I.E + Dissociating energy + E.A = Lattice Energy Heat of formation= Dissociation Energy + Sublimation Energy + Ionization Energies – Electronic Affinities + Lattice Energy. The value of this energy is the difference of values from Step 1 and Step 4. Now, the metal and nonmetal will be combined to release an energy called lattice energy. Following that, subtract the electron affinity of nonmetal from the value of step 3. To do this, add the ionization energy of the metal to the value of step 3. Calculate the dissociation energy for the nonmetals and add them to the value obtained in step 2.īoth metals and nonmetals are now to be changed to ionic solids. Metals usually exist as single atoms, which is why there won't be any dissociation energy for them. Add the enthalpy change by turning the elements into the gaseous states. This will be used as the energy of ionic solids in the equation.Īccording to the Born–Haber cycle, the elements shall be in gaseous forms. Subtract the heat of formation of the ionic solid by combining these elements. The Born – Haber cycle is based on Hess' law and its application of ionic solid.ĭetermine the energy of all the metals and nonmetals in the elemental forms. Li(s), ->, Li(g), energy of sublimation, +161 kJ, endothermic 2. The overall change in energy is determined by breaking the process into steps and adding changes in each step. Lattice Energy Calculations via Born-Haber cycle 1. 
It can be either positive or negative based on the atoms involved and their interaction. It is the change in energy when a particular compound is formed from elements. It is again an input of energy and is always positive. Known as the energy of atomization, it is required to turn the compound from solid to gas while passing through the liquid phase. The magnitude of it is dependent on the electronegativity of the atoms involved in the compound. Dissociation is an endothermic process that requires an input of energy. The energy needed to break a compound apart. BornHaber cycle applies Hesss law to calculate the lattice enthalpy by comparing the standard enthalpy change of formation of the ionic compound (from the. The electron affinity is known for increasing from left to right and decreasing from top to bottom in a periodic table. When used to calculate the lattice energy, one has to subtract the electron affinity and not add it because of the positive value. The energy released is known to have a negative value, but due to the definition of affinity, it is denoted as a positive value. The energy released when an electron is added to a neutral ion/atom. However, there are some exceptions due to the unpredictable stability of completely filled/half-filled orbitals. Ionization energy is noted to be increasing across the periodic table from left to right and decreasing from top to bottom. In order to do this, there should be an input of energy, which is why it is always positive. The energy required to take off an electron from the neutral ion/atom. There are several other concepts that one has to understand before getting to the Born – Haber cycle and applying it to determine the lattice energy of an ionic solid. A Born-Haber cycle applies Hess' law to calculate the lattice energy by comparing the standard enthalpy change in the formation of the ionic solid from the elements to the enthalpy that is required to form the gaseous ions from the elements. 
It helps us to calculate the lattice enthalpy of an ionic compound. The lattice energy is the enthalpy change involved in the formation of ionic solids from gaseous ions or the energy involved in breaking the ionic solids into gaseous ions. The Born-Haber cycle represents a series of steps leading to the formation of an ionic compound from its constituent gaseous ions. The cycle is mostly concerned with the formation of an ionic solid from the metals (Group I or Group II) when reacted with a halogen or a non-metallic element like oxygen.īorn-Haber cycles are primarily used in calculating the lattice energy, which cannot be measured otherwise. The Born-Haber cycle is a classic approach to measure the reaction energies. 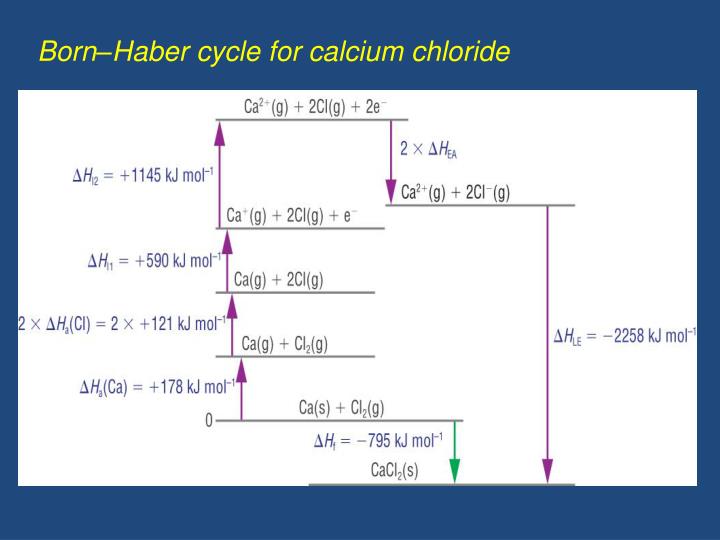
In order to understand the idea of lattice energy and calculate it, a Born-Haber cycle is used. This is where the Born-Haber cycle allows us to determine and understand the lattice energies of the ionic solids. The lattice energy present cannot be measured. These compounds also have additional stability because of the lattice energy present in their solid structure. The enthalpies required for the formation of the ionic molecules do not account for stability alone. The n values and the electronic configurations (e.c.Ionic solids are stable compounds. Where N is the Avogadro's number (6.022x10 -23), and n is a number related to the electronic configurations of the ions involved.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
